
Mottled Sculpin, Cottus bairdi

Mottled Sculpin, Cottus bairdi
The mottled sculpin, Cottus Bairdi, is a small (7-10 cm) benthic fish that lives in the shallower areas of lakes and streams. Field studies have discovered that the mottled sculpin preferably feeds at night, presumabely to avoid becoming a tasty morsal for large daytime hunters. Because it feeds in low-light conditions, sculpins must have an effective way of detecting food without using their eyes. Studies over the last 20 years have determined that sculpins use a specialized hair cell sensory system, the mechanosensory lateral line, to detect differences in water pressure that occur along the length of their bodies as prey animals swim by.
Prey capture behavior in mottled sculpin is characterized by a series of orienting "hops" along the substrate of its environment. These hops are made in such a way that it allows the sculpin to move toward the prey item (source of vibration) and at the same time orient their body so that their mechansonsory receptors are in the best place to help locate the prey, thus increasing the likelyhood of a successful strike. The diagrams below show two stereotypical pathways taken by a sculpin on its way to a prey item. In these diagrams the arrowhead represents the sculpin with the snout at the tip. Notice that during its approach to the prey (a vibrating bead at the center of the tank), the sculpin does not orient itself directly toward the prey item or perpendiular to the axis of water pressure (dotted lines) until it is ready to strike (position 6 or 7).
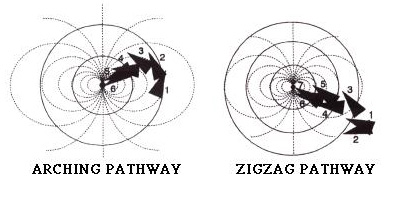
Fig. modified from Coombs and Conley,1997.
Central Nervous System: Thus far the neural substrates involved in this system have been studied more extensively in other animals, but the primary brainstem nucleus for mechanosensory processing is assumed to have the same characteristics in sculpin as in other animals. The medial octavolateralis nucleus (MN) has 3 distinct layers (see diagram below): An overlying molecular layer consisting of parallel fibers running from the cerebellum and and other areas, a primary cell layer which has large multipolar neurons that receive information from both the molecular layer and primary receptor afferents and then project to higher brain centers, and the deep neurpil where primary receptor afferent fibers synapse onto the projection cells either directly or disynaptically via inhibitory interneurons (small black cell).
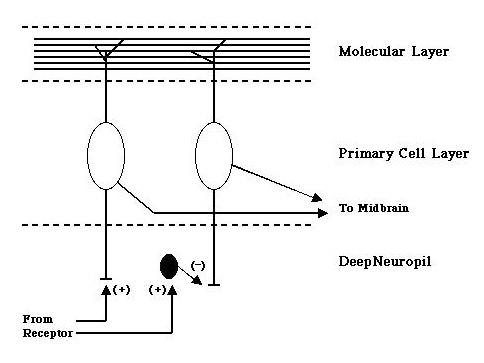
Little is known as of yet how this nucleus processes mechanosensory information, but recordings from projection cells have suggested that the receptive fields of these cells may show a form of center surround antagonism.
Periphery: Recordings from the primay receptor afferents in sculpin have shown that the mechanosensory receptors, known as neuromasts are capable of encoding information about pressure differences across different receptors. These studies have led to the development of a model, diagrammed below, of how the fish might localize a prey source with its mechanosensory system.
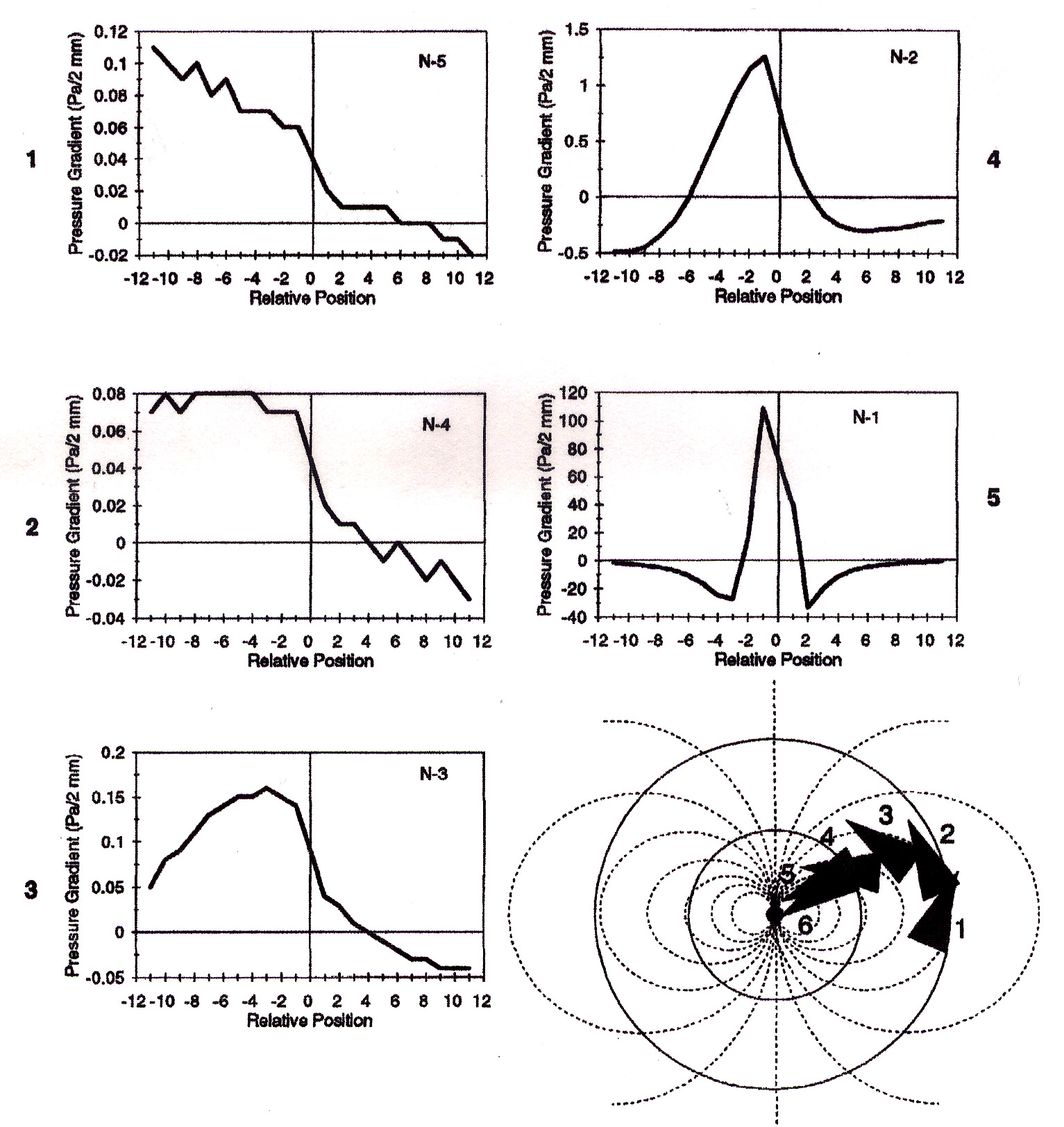
Y-Axis = presure gradient (Pa/2mm), X-axis = relative postition of
neuromasts with respect to the fishes snout (position "0"). Put
simply the "-2" on the x-axis represents the second neuromast to the
left of the snout while "+2" represents the second neuromst to the
right of the snout. Numbers to the sides of each plot correspond to
the fishes position in the diagram of the pathway. Fig. from Coombs
and Conley, 1997b.
In the model, as the fish approaches the source of vibration, the corresponding plots show how the differences in pressure might "appear" to the fish. Using these cues, each orientation step is used to get nearer the prey item and also to bring the peak pressure gradient in line with its mouth facilitating a successful strike.
Early classics:
Dijkgraaf, S (1963) The functioning and significance of the
lateral-line organs. Biological Reviews 38: 51-105
Hoekstra, D, Janssen, J (1986) Lateral line receptivity in th mottled sculpin (Cottus bairdi). Copeia 1986:91-96.
A Little Later:
Janssen J, Coombs S, Hoekstra D, Platt C (1988) Anatomy and
differential growth of the lateral line system of the mottled
sculpin, Cottus bairdi. Brain, Behav. Evol.
30(3-4): 210-229.
Coombs S, Janssen J (1990) Behavioral and neurophysiological assessment of lateral line sensitivity in the mottled sculpin, Cottus bairdi. J. Comp. Physl. A 167(4): 557-568.
Some Recent Advances:
Coombs S, Conley RA (1997a) Dipole source localization by mottled
sculpin I: Approach strategies. J. Comp. Physl. A
180:387-399.
Coombs S, Conley RA (1997b) Dipole source localization by mottled sculpin II: The role of lateral line excitiation patterns. J. Comp. Physl. A 180:401-415.
Conley RA, Coombs S (1998) Dipole source localization by mottled sculpin III: Orientation after site-specific, unilateral denervation of the lataral line system. J. Comp. Physl. A 183:335-344.
Coombs S, Mogdans J, Halstead M, Montgomery J (1998) Transformation of peripheral inputs by the first-order lateral line brainstem nucleus. J. Comp. Physl. A, Sensory, Neural, and Behavioral Physiology May '98; 18(5):609-626.
Coombs S (1999) Signal detection theory, lateral-line excitation patterns and prey capture behavior of motteld sculpin Animal Behavior Aug '99; 58(2):421-430.
