

The parasitoid fly, Ormia ochracea is native to Florida and other parts of the southeastern US. It is quite small, measuring at most 1 cm in length.
Females of the species parasitize cricket species of the genus Gryllus, upon which they lay 1st instar larvae. The larvae burrow into the body of the cricket where they feed and moult for about 7 days after which they emerge from the cricket and pupate. Crickets do not usually survive long after emergence, possibly due to toxicity effects of the larva purging its gut before leaving its host.
Unlike most predatory or parasitoid fly species, Ormia do not use vision to track their targets. Instead, Ormia females use their remarkable hearing abilities to locate singing crickets (usually at night) on which to deposit their larvae. Using loudspeaker playing recorded cricket calls, researchers have shown that female flies can locate crickets from several meters away with high degrees of accuracy (within a few cm of the center of the sound source). If the song is interrupted while the fly is tracking the sound in-flight, the fly can still gauge both distance and direction from previous information to find the source.
The flies hear using two very small (~1 sq. mm area) ears, located on the front of their thorax, just below where the head/neck attaches - also called the prosternum. The ears are composed of two flexible tympanal membranes joined and linked by a small exoskeletal structure, the presternum. In the center of the tympanal membranes are tiny tympanal pits, where the auditory nerve carrying sensory information from each ear attaches to the exoskeleton. The whole organ is less than 2mm wide. Unlike similar ear structures in other insect species, the ears in Ormia are sexually dimorphic - with male flies possessing smaller tympanal membranes, and a generally reduced ear structure. It is also believed that these smaller ears lack some of the sensitivity to cricket song that female flies possess.
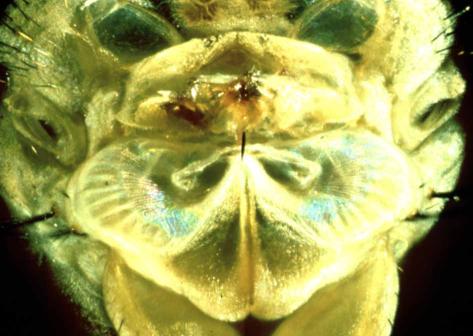
Inside the organ lies an internal air sac, filling most of the prosternum. The auditory organs (which transduce the vibration of the tymapanal membranes into neural signals passed on to the fly's brain) are the bulba acustica (BAc). One end of the organs is attached to the tympanal pits, the other end to internal exoskeletal attachment points. Each BAc has between 100-110 sensory neurons, which project to the auditory nerves that leave the rear of the organs and project to the thoracic ganglia in the insect's central nervous system.
Something that initially puzzled researchers was how such precise hearing ability could arise from such a small ear structure. Most animals with true ears detect and locate sound sources using two sound features: the Interaural Time Difference (ITD) and the Interaural Level Difference (ILD).The ITD is the difference in the time it takes sounds to reach each ear - if a sound is to our left, it will reach our left ear fractions of a second before it will reach out right ear. If a sound is directly ahead of us, it will reach both ears at the same time. The ILD is the difference in sound intensity measured between both ears. Again, if a sound is to our left, our left ear will receive a slightly louder sound than our right. This is because the sound loses some of its itensity as is travels through both the air and our heads as it moves the further distance to our right ear.
The extremely small size of Ormia's ears presents a problem in using both the ITD and ILD to localize a given sound. The tympanal membranes of the fly are approximately 500 micrometers (~0.5 mm) from each other, the time it takes for a sound to pass past one ear, and then the other is very small. At maximum (a sound coming completely from either the left or the right), the ITD would be just 1.5 microseconds (compared to 500-700 microseconds in humans). In addition, the ILD measured between the ears would be less than 1 decibel, and often there would be no measurable difference at all! Given these limitations, it should be extremely difficult, or almost impossible for the female flies to locate singing crickets on which to lay their larvae, yet they manage to locate hosts both in the wild and in the lab! Through careful study, biologists have discovered that Ormia have both mechanical and neural tricks up their sleeves that they can use to turn the tiny sound location cues into signals large and strong enough for their nervous systems to successfully locate the sound's source.
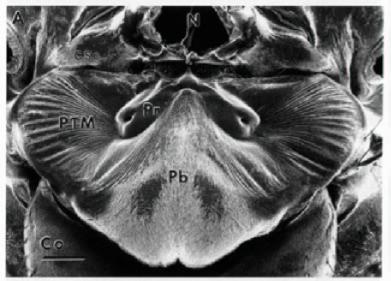
In studying the mechanical nature of the fly's ears, researchers found that the presternum structure linking both tympanal membranes plays a critical role in sound detection and localization. The structure acts as a flexible "lever" and helps transfer and amplify vibrational energy between both tympanal membranes. As the sound hits both membranes with slightly different phases and amplitudes, the presternum sets up asymmetrical vibrational modes in each membrane, through both "bending" and "rocking" actions. The net effect is to increase the asymmetry of the membrane vibrations, to make it easier for the nervous system to distinguish on which side the sound is coming from.
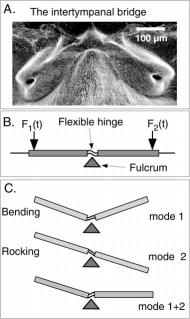
With the help of the presternum acting as an intertympanal bridge, the ITD is increased from 1.5 us to about 55 us, and the ILD is increased from less than 1 decibel to over 10 decibels. As far as biologists have studied - this mechanism for amplifying asymmetries in auditory signals is unique in the animal kingdom showing that big things really CAN come from small packages!
In addition to mechanical mechanisms to facilitate auditory localization, the auditory nervous system in Ormia is also adapted to help it find crickets from their calls. Researchers looked at the responses of neurons leading from the bulba acustica to the CNS and their electrical response properties to stimulation with a range of sound intensities and frequencies. They classified 3 types of auditory afferents according to their consistent response properties:
The neurons responded most strongly to sound frequencies in the range of 4-9 kHz, which includes the frequencies present in host cricket songs. Interestingly, no neurons were found that had their strongest response at 4.5 kHz - which is the strongest frequency in the Gryllus host song. Regardless of type, all observed neurons had an inverse intensity/latency relationship - the stronger the stimulus, the shorter the time until the neuron begins to respond.

But what does the above have to do with how the neurons encode the sound direction? The inverse intensity/latency properties, single-spike responses seen in most receptors, and low variability in timing could indicate that the relative timing of spikes could be used to encode sound source direction. Comparing the different latencies of incoming spikes between both hearing organs can indicate from what direction an sound originates. But, the low ITD presented to the system (10-50 us) is encoded by receptors with an average variability in timing precision of 70 us! The system can still code accurate timing information with less accurate and thus very variable receptors by using a population of receptors. Similar characteristics are seen in other systems that must encode precise timing information. Precise temporal encoding using a population of less accurate receptor neurons is known as temporal hyperacuity.
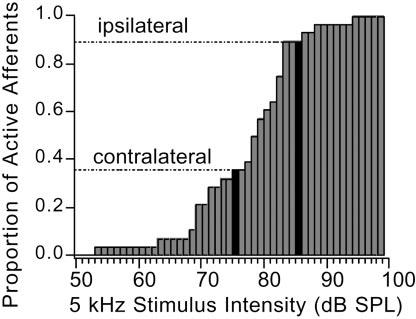
In addition to using relative timing, Ormia may also use the differences in the sound intensity thresholds in the population of auditory receptor neurons to encode sound direction. Researchers have found that the BAc auditory receptors have a broad distribution of intensity thresholds, from 50-96 dB. Depending on what direction a sound originates, it will excite different proportions of receptors on each side of the fly due to the different intensities perceived. Of course, neither latency encoding nor population size encoding are mutually exclusive - it is quite possible that BOTH mechanisms are present in the Ormia CNS. Further neurophysiological and behavioral research will shed light on the actual mechanisms present.
The fact that individual Ormia females can localize prey using an extremely compact, yet accurate auditory system suggests the possibility of designing and building sound amplifying and detecting devices on a much smaller scale than currently used. The underlying guiding principle is that over the course of millions of years, organs and organ systems have evolved with high degrees of specialization and efficiency, and that some of their characteristics and functional principles may be of use in some engineering applications. Currently, several researchers and collaborators are working on "biomimetic" sound devices that use principles found in Ormia ochracea.
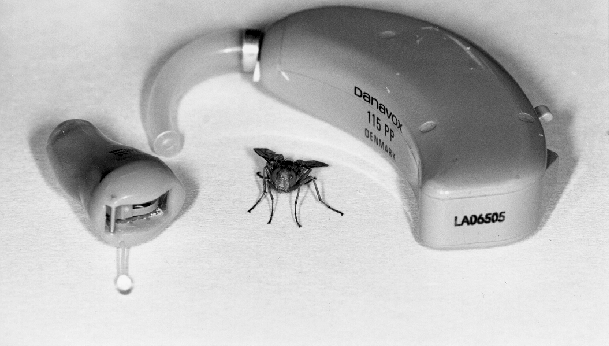
Dr. Hoy's lab at Cornell, along with Dr. Miles's group at SUNY Binghamton, Dr. Mason's lab at the University of Toronto at Scarborough, and collaborating engineers are working to fabricate a directional microphone that builds on the physical principles that the Ormia tympanal membrane uses to amplify ITD and ILD differences. The current designs use MEMS (Micro-Electro-Mechanical Systems) fabrication techniques, which are similar to the way silicon computer chips are designed and manufactured. Such a device could be used to precisely measure sound intensity of very low-intensity sounds and conduct measurements with high degree of accuracy. It may also be used as a directionally sensitive microphone that would be both small in size and low in cost. The most ambitious application would be to use the technology to develop an advanced hearing aid. With directional sensitivity, the aid would preferentially amplify sound coming from one direction, such as the front, from which the user would hear most speech. Current hearing aids amplify ALL sound, both speech coming from one direction, and ambient noise from all the others. Directional selectivity would greatly help the hearing-impaired understand conversation in noisy environments. Currently, such microphones are too large to be used in hearing aids that lay in the ear canal. But with technology developed from Ormia, that size could be reduced to only 2mm across.
Hoy lab - Many researchers using Ormia got their start in Dr. Ron Hoy's lab at Cornell. Dr. Hoy also studies stalk-eyed flies, katydid bushcricket acoustic communication, and other features in insect sensory biology. Several of the photographs on this web page are from his web site.
Miles lab - Dr. Miles studies Ormia as well as the system's application to hearing aid and microphone technology. Also featured on this site is a movie demonstrating how the fly' directional abilities were tested.
Mason lab - An alum from the Hoy lab, Dr. Mason studies Ormia neurophysiology and animal communication. His lab web site also offers videos of Ormia in action, and the site includes many interesting photographs of the flies (as well as other insects Dr. Mason studies), including pictures of experimental preparations.
Laboratory of Bioacoustics and Sensory Biology at the University of Bristol - Currently the home of Dr. Daniel Robert, research here also focuses on Ormia auditory physiology, biomechanics, and the creation of biomimetic acoustic sensors.
