
Spiny lobster, Panulirus interruptus, photo: James Murray

Spiny lobster, Panulirus interruptus, photo: James Murray
The lobster is a crustacean, also classified as an arthropod, and is a very popular experimental invertebrate. Its experimental appeal comes from what we would consider poor manners. The lobster does not chew its food orally, instead it swallows its meal whole and allows its stomach do to the chewing. It was discovered that a particular ganglion, the stomatogastric ganglion (STG), controls the rhythmic contractions of the stomach and gastric teeth.
Tree of Life: Decapoda
The overt observation of the digestive behavior of the lobster
is not immediately accessible. Until we have transparent lobsters, there
are two ways that neuroethologists can study the digestive behavior of this
crustacean. 1) They can use endoscopy to monitor and record the
intact workings of the digestive tract (it's a little dark, but
effective). 2) They can dissect out the relevant ganglia and motor
nerves and record from these neurons and nerves in the dish (or in vitro
).
The first method using endoscopy was necessary at the beginning of these
studies to confirm that what was occurring in the dish was physiologically
relevant. Once that relevance was established, most researchers just relied
on their recordings and observations in the dish to constitute what is known
as "fictive" behavior; e.g. the behavior that would have occurred
when certain nerves etc. fire, except that the rest of the musculature and
structure has been removed.
The behavior of the lobster digestive system is decidedly rhythmic. However,
it is not similar to the rhythmicity that is found in vertebrate digestive
systems. Unlike other animals, lobsters stomachs are ectodermal tissue controlled
entirely by striated muscles. No movement occurs in the stomach unless a
direct message comes from the motoneuron of the STG. This rhythmic behavior
of the muscles, and stand alone control of the STG in the dish highlights
that the stomatogastric nervous system in the lobster contains a central
pattern generator.
A central pattern generator (CPG) is a mechanism
that controls a cyclically repeated behavior, usually walking, flying, chewing;
that neuronal mechanism resides within the central nervous
system (in this case the STG). Although the pattern can be influenced by
peripheral cues and modulation, the ultimate control of the pattern lies
within the CPG.
There are several rhythms active in the lobster digestive system, and these
correspond to the organs involved with the behavior. There is an esophageal
rhythm that allows the lobster to take in food. There is the cardiac rhythm
that involves the cardiac sac, the place where food is stored in the lobster.
In addition to these two rhythms there are two more that have been well
characterized for the lobster. The first of these rhythms is the gastric
rhythm. This rhythm is responsible for the control of the calcified
teeth that lay within the gastric mill of the lobster. Food moves from the
cardiac sac into the gastric mill and is macerated by the action of the
lateral and medial teeth. The second well characterized rhythm is the pyloric
rhythm. The pyloric rhythm controls the pylorus or pyloric filter
which is a system of valves and sieve plates that helps sort out the food
particles so that they may be further digested. Each of these rhythms involves
coordinated contractions of groups of muscles, sometimes firing together,
and at other times firing in opposition. This rhythm generation has been
scrutinized at the cellular level, and that research has generated excellent
diagrams of the neural circuitry.
The neural circuitry controlling the gastric rhythm is well characterized
at the cellular level. All of the neurons involved in controlling the movements
of the lateral and medial teeth have been identified, and their connections
have been mapped. The gastric rhythm produces a coordinated chewing movement
in the gastric mill that is controlled by a CPG. The CPG has 10 motoneuron
and one interneuron at its disposal to control the gastric mill. There are
quite a few muscles, called gm muscles, that are responsible for
controlling the calcified teeth. These muscles are directly innervated by
the motoneuron of the STG
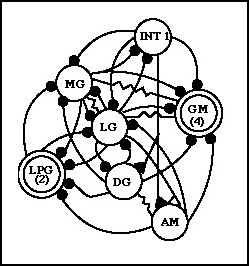
Gastric Rhythm Circuitry
INT 1: interneuron 1
MG: medial gastric neuron
LG: lateral gastric neuron
DG: dorsal gastric neuron
AM: anterior median neuron
GM: gastric mill neurons
LPG: lateral posterior gastric neurons
The black filled circles (small) represent inhibitory chemical synapses,
and the resistor shaped connections represent direct electrical coupling
between cells.
redrawn and modified from Marder and Calabrese, 1996
The summary of neural activity: The LG neuron is responsible
for pulling the lateral teeth apart, as is the MG
neuron. These neurons control muscles gm5b and gm 6b and gm9 respectively.
Food is moved between the lateral teeth at this point, most likely through
coordination with the cardiac sac. Next the LPG neurons
fire, and these cause the teeth to close together. The LPG neurons control
muscle(s) gm3c. The antagonist neurons to the first two sets are now activated.
The GM neurons cause the medial tooth
to move forward and downward, scraping across the food held by the lateral
teeth. These neurons control muscles gm1,2 &;3. Then the DG and
AM neurons reset the position of the medial tooth and constrict
the cardiac sac. They control muscles gm4 and c7. At the end of this cycle
the whole behavior begins again in the same stereotyped cyclical pattern.
Unlike some CPG's there is no one driving cell within the network. Therefore
the gastric mill CPG is considered an emergent network. The alternating
rhythmicity of the network is controlled not only through the electrical
coupling and inhibitory connections, but also through phenomena that are
occurring at the biophysical level such as post-inhibitory rebound, IA and
IH currents, and plateau potentials.
The neural circuitry controlling the pyloric rhythm is more fully characterized than that of the gastric rhythm. The pyloric rhythm is responsible for sorting and sifting the food particles that have been ground up by the gastric mill. This circuit has 13 motoneuron in it and one interneuron. Several of the neurons, the PD and the PY are represented in multiple copies, thus simplifying the circuit at some level. A great deal is known about the chemical nature of the synapses in the pyloric circuit and that has increased our understanding of factors that affect individual neurons in an oscillating circuit.
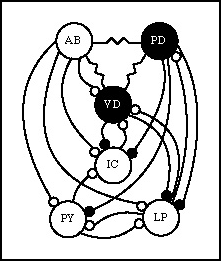
Pyloric rhythm circuitry
AB: anterior burster neuron
PD: pyloric dilator neurons (2)
VD: ventricular dilator neuron
IC: inferior cardiac neuron
LP: lateral pyloric neuron
PY: pyloric neurons (8)
Small circles represent inhibitory synapses, black filled cells and synapses
are cholinergic, and empty cells and synapses are glutamatergic. Resistor
shaped connections indicate electrical connections directly between cells.
The summary of neural activity: There are three main phases of activity
in the pyloric circuit. The AB neuron serves as the pacemaker
for this circuit. Because of its electrical coupling to the AB neuron, the
PD neuron also fires with the AB pacemaker cell and helps
to establish the rhythm. The AB/PD neurons strongly inhibit the other cells
in the network. When AB/PD fire they open a valve from the gastric mill
to the pyloric region. They synapse on the pyloric dilator muscle(s).
This allows food particles to flow into the pylorus. The LP neuron
is the first neuron to recover from the inhibition, and it fires, causing
the valve to close. The IC cell fires in phase with the
LP cell, also aiding in control of the valve. The LP cell synapses on the
lateral pyloric muscle. The LP neuron also inhibits the AB/PD neurons which
prevents them from firing again. In the final phase of the cycle the PY
neurons recover from inhibition and begin firing. These neurons
control the pyloric muscles themselves that are responsible
for moving the sieve plates that sort out the food for further digestion.
The VD cell often fires in phase with the PY cells, but
its firing is complex and not easily correlated to just one movement.
Again, as in the gastric rhythm, the activity of the network is controlled
by many factors, including the endogenous bursting activity of the AB neuron,
inhibitory connections, and the biophysical properties of the individual
cells.
Modulation
A discussion of the lobster STG would be incomplete without briefly mentioning
some of the most exciting findings in this system. It was discovered that
bathing the lobster STG prep (and preps in other crustaceans) with neuromodulators,
substances like octopamine, dopamine,
and serotonin, can cause dramatic shifts in the phasing
and connections between the neurons in the CPG.
It was once believed that CPG's were 'hard wired' for a specific function,
and each function would have its own CPG in the CNS. However, with these
results from the lobster it now appears that phenomena like neuron switching,
phase shifting, and strengthening (or weakening) of connections are quite
routine.
This causes scientists to reformulate their definitions for what a CPG really
is, not something 'hard wired' but something much more flexible than that.
In retrospect, this intuitively makes sense since many of the same neurons
control the same muscles that animals use for walking, running and cantering.
Research is currently being done using a variety of neuromodulators and
neurohormones to test their effects on this system. Already tens of compounds
have been found to have an effect. In addition, researchers are looking
for the endogenous compounds that may be released by neurons outside of
the CPG itself, to determine the physiological conditions in which the modulators
are released.
Early classics:
Hartline, D.K. Pattern generation in the lobster (Panulirus) stomatogastric
ganglion. II. Pyloric network stimulation. Biol. Cybern. 33:223-236,
1979.
Hartline, D.K. and D.V. Gassie. Pattern generation in the lobster (Panulirus ) stomatogastric ganglion. I. Pyloric neuron kinetics and synaptic interactions. Biol. Cybern. 33:209-222, 1979.
Mulloney, B., and A.I Selverston .
Organization of the stomatogastric
ganglion in the spiny lobster. I. Neurons driving the lateral teeth. J.
Comp. Physiology.
91:1-32, 1974.
Mulloney, B. and A.I Selverston. Organization of the stomatogastric ganglion in the spiny lobster. II. Neurons driving the medial tooth. J. Comp. Physiology. 91:33-51, 1974.
Mulloney, B., and A.I. Selverston. Organization of the stomatogastric
ganglion in the spiny lobster. III. Coordination of the two subsets of the
gastric system.
J. Comp. Physiology. 91:53-78, 1974.
Reviews:
Marder, E. and R. Calabrese. Principles of rhythmic motor pattern generation.
Physiol. Reviews. 76:687-717, 1996.
Selverston, A. Modulation of circuits underlying rhythmic behaviors. J. Comp. Physiology. 176:139-147, 1995.
Recent Developments:
Harris-Warrick, R.M., L.M. Conigilo, R.M. Levin, S. Gueron and J. Guckenheimer.
Dopamine modulation of two subthreshold currents produces phase shifts in
activity of an identified motoneuron. J. of Neurophysiology.
74:1404-1420, 1995.
Schneider, H., P.Budhiraja, I.Walter, B.S. Beltz, E. Peckol, and E.A.
Kravitz. Developmental expression of the octopamine phenotype in the lobsters,
Homarus americanus. J. of Comp. Neurology. 371:3-14,
1996.
If you are interested in learning more about the lobster or the crustacean field of research there are several great web sites to visit. The crustacean research community is well connected, and their web sites are informative as well as a great jumping point for further reading.....
Dan Hartline's STG Home Page, a great starting point with tons of pictures and data.
The Marder and Abbott Neuroscience Group, another good link from big names in the field.
Robert Huber's Central Terminal for Crustacean Neuroscience, you're bound to find something interesting here!
Robert Huber's Crusties Neuroethology
CRAYFISH - UCSD , Al Selverston's lab.
